 It is sold as an immediate-release product for moderate to severe pain. It is used for managing moderate to severe pain. The first oxycodone containing-drug, Percodan, was approved by the FDA in 1950. Percocet or oxycodone is a semisynthetic opioid analgesic derived from thebaine and produced in a lab. For instance, Percocet is derived from thebaine, while Norco is derived from codeine. Percocet and narco have multiple similarities and differentiate only on their sources. Drug Enforcement Administration (DEA) due to a high potential for abuse and addiction. Percocet and Norco are in the same drug class and designated as schedule II drugs by the U.S. Opioids are addictive, and many drug abusers use them for the euphoric feeling they provide. Opioids, also known as opiates, narcotics, and painkillers, include prescription and illegal drugs derived from or mimic the natural substances found in the opium poppy plant. If you are having difficulty accessing this website, please call or email us at 1-85 or so that we can provide you with the services you require through alternative means.Percocet (generic name oxycodone) and Norco (generic name hydrocodone) are both opioid prescription drugs used for managing moderate to severe pain. GoodRx works to make its website accessible to all, including those with disabilities. Truven Health does not assume any responsibility or risk for your use of the Truven Health products. ALL IMPLIED WARRANTIES OF MERCHANTABILITY AND FITNESS FOR A PARTICULAR PURPOSE OR USE ARE HEREBY EXCLUDED. Additionally, TRUVEN HEALTH MAKES NO REPRESENTATION OR WARRANTIES AS TO THE OPINIONS OR OTHER SERVICE OR DATA YOU MAY ACCESS, DOWNLOAD OR USE AS A RESULT OF USE OF THE THOMSON REUTERS HEALTHCARE PRODUCTS. Truven Health and GoodRx make no representation or warranty as to the accuracy, reliability, timeliness, usefulness or completeness of any of the information contained in the products. These products are provided 'AS IS' and 'as available' for use, without warranties of any kind, either express or implied. The use of the Truven Health products is at your sole risk. Cold Spring Harbor Perspectives in Medicine. Neonatal Opioid Withdrawal Syndrome (NOWS): A transgenerational echo of the opioid crisis. Opioid analgesic Risk Evaluation and Mitigation Strategy (REMS). FDA Drug Safety Communication: FDA warns of rare but serious skin reactions with the pain reliever/fever reducer acetaminophen. Hydrocodone bitartrate and acetaminophen solution. Opioid use and opioid use disorder in pregnancy. The American College of Obstetricians and Gynecologists. Risk Evaluation and Mitigation Strategy (REMS). Symptoms and causes of adrenal insufficiency & Addison's disease. National Institute of Diabetes and Digestive and Kidney Diseases. Adrenal insufficiency and Addison's disease. 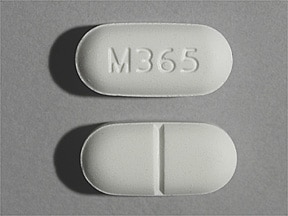
Comparison of oxycodone and hydrocodone for the treatment of acute pain associated with fractures: A double-blind, randomized, controlled trial. Cognitive and behavioral impact on children exposed to opioids during pregnancy. Hydrocodone bitartrate and acetaminophen tablet. 
National Institute of Child Health and Human Development.Įywa Pharma. Centers for Disease Control and Prevention.ĭrugs and Lactation Database (LactMed). CDC clinical practice guideline for prescribing opioids for pain – United States, 2022. If both medicines are prescribed together, your doctor may change the dose or how often you use one or both of the medicines.ĭowell, D., et al. Using this medicine with any of the following medicines may cause an increased risk of certain side effects, but using both drugs may be the best treatment for you.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
